Comment:
Another study (see here) showing that PPIs lead to worse outcomes, this time reducing both progression-free and overall survival. We have better tools, but if a medication is necessary, other options such as H2 blockers are a better choice.
Summary:
Clinical Bottom Line
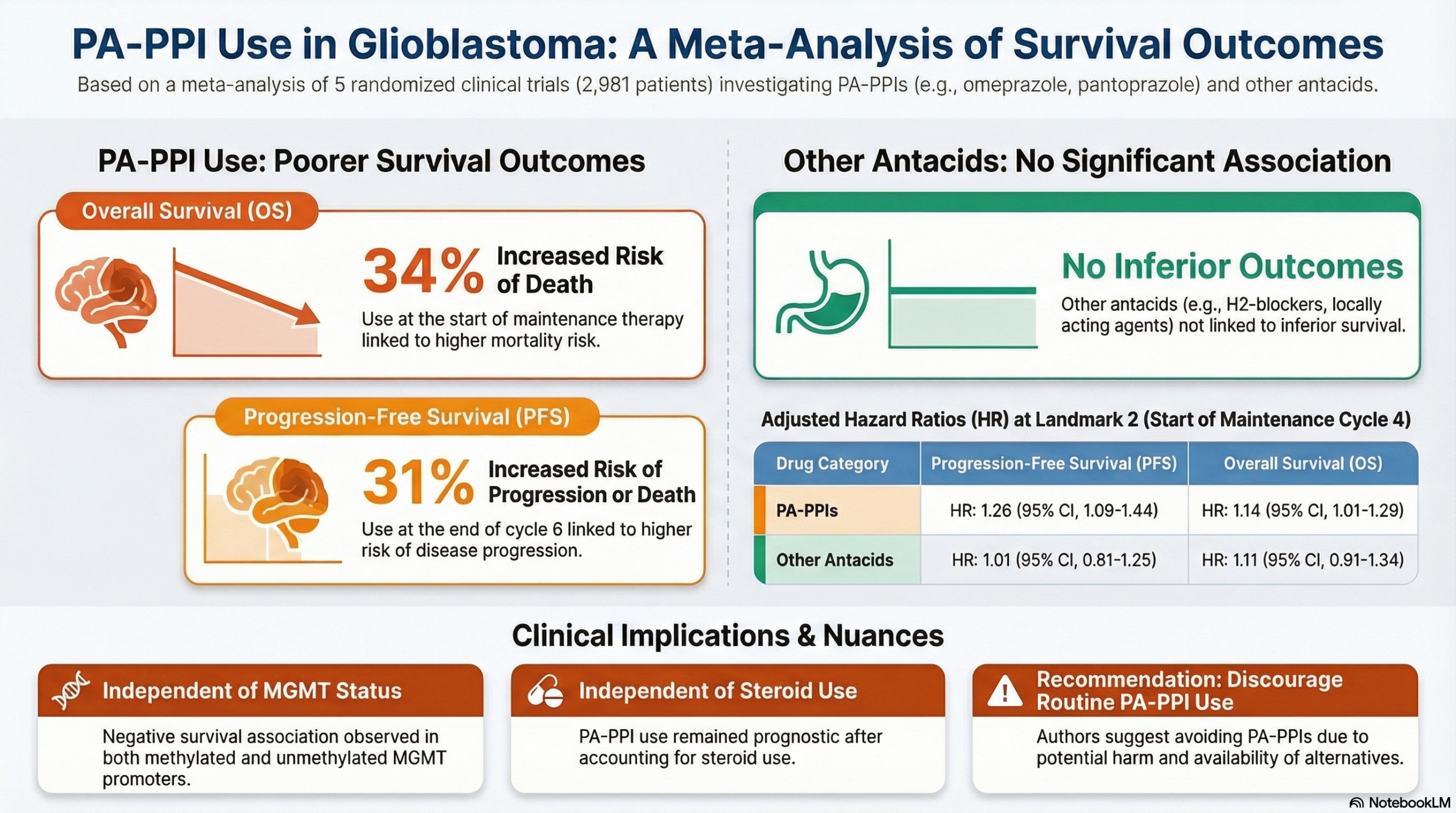
This meta-analysis of individual patient data suggests that the use of specific Proton Pump Inhibitors (PPIs)—specifically those that are potent activators of ALDH1A1 (PA-PPIs) like omeprazole and pantoprazole—is associated with significantly inferior Overall Survival (OS) and Progression-Free Survival (PFS) in patients with newly diagnosed glioblastoma. Notably, this negative association was not observed with other antacid medications (such as H2 blockers or locally acting antacids).
While the data are derived from high-quality Randomized Clinical Trials (RCTs), the comparison regarding PPI use is observational. Therefore, while causation cannot be definitively proven due to potential confounding by indication (e.g., sicker patients requiring more steroids and thus more PPIs), the specificity of the finding to PA-PPIs and the availability of safe alternatives suggests that clinicians should consider avoiding PA-PPIs in this population.
Results
Summary of Results: The study analyzed data from 2,981 patients across 5 RCTs. The authors utilized a landmark analysis approach to account for drug exposure over time.
-
Primary Outcome (Survival): The use of PA-PPIs was associated with worse survival outcomes compared to no drug use.
-
Overall Survival (OS): Adjusted Hazard Ratio (HR) was 1.34 (95% CI, 1.08-1.66) at Landmark 1 (start of maintenance cycle 1) and 1.14 (95% CI, 1.01-1.29) at Landmark 2. This indicates a 34% and 14% increase in the risk of death, respectively, for patients taking PA-PPIs.
-
Progression-Free Survival (PFS): Adjusted HRs showed a consistent negative association at all measured landmarks: HR 1.14 at Landmark 1, HR 1.26 at Landmark 2, and HR 1.31 at Landmark 3.
-
Specificity of Agent: No statistically significant association with inferior survival was found for other antacid drugs (weak ALDH1A1 activators, H2 blockers, or local antacids) after multivariate adjustment.
-
Independence of Effect: The negative association of PA-PPIs was observed independently of MGMT promoter methylation status and steroid use.
Assertive Critical Appraisal
Certainty of Evidence (GRADE Framework)
-
Rating: Low to Moderate.
-
Downgrading Factors: Although the data comes from RCTs, the analysis of PPI use is observational. Patients were not randomized to PPIs; they received them as concomitant medication. This introduces the risk of confounding variables that cannot be fully adjusted for, despite the use of multivariate models.
Risk of Bias & Confounding
-
Confounding by Indication: This is the most significant threat to validity. Patients with larger tumors or more severe edema require higher doses of steroids, and consequently, are more likely to be prescribed PPIs for gastric protection. These patients inherently have a worse prognosis.
-
Authors’ Mitigation: The authors attempted to control for this by adjusting for “steroid use (yes/no)” and performance status in their multivariate models.
-
Critique: The adjustment for steroid use was binary (yes/no). The study did not capture overall steroid dose or duration. Therefore, residual confounding is likely—patients on PA-PPIs might have been on higher doses of steroids than those on other antacids, reflecting more aggressive disease. However, the fact that other antacids did not show the same negative survival signal strengthens the authors’ hypothesis that the effect is drug-specific rather than solely due to the underlying need for gastric protection.
Biological Plausibility The study proposes a specific mechanism: PA-PPIs (omeprazole, pantoprazole) activate ALDH1A1, an enzyme linked to cancer stemness and protection against oxidative stress/chemotherapy. This provides a biological rationale for why these specific drugs—and not other antacids—would be detrimental.
Reporting Quality Assessment (PRISMA)
-
The study adhered to PRISMA guidelines.
-
Data Handling: The use of landmark analysis is a strength, as it avoids “immortal time bias” (a common error where treatment responders simply live longer to receive the drug).
-
Missing Data: The authors rigorously handled missing data, utilizing multiple imputation when missingness exceeded 5% but was less than 15%.
Applicability The findings are highly applicable. The study population reflects a standard cohort of newly diagnosed glioblastoma patients eligible for clinical trials. The alternatives to PA-PPIs (H2 blockers like famotidine or weak ALDH1A1 activators like lansoprazole) are widely available and inexpensive, making the clinical pivot low-risk and potentially high-reward.
Research Objective
To explore the associations of the use of potent ALDH1A1-activating PPIs (PA-PPIs) and other antacid drugs with survival outcomes (PFS and OS) in patients with newly diagnosed glioblastoma.
Study Design
-
Design: Meta-analysis of individual participant data (IPD) from 5 prospective Randomized Clinical Trials (ACT IV, AVAglio, CENTRIC, CORE, EORTC 170920).
-
Exposure Assessment: Drug use assessed at baseline and three specific landmarks (start of maintenance cycles 1 and 4, and end of cycle 6).
-
Statistical Analysis: Multivariate Cox proportional hazards models adjusting for age, sex, performance status, steroid use, extent of resection, and MGMT status.
Setting and Participants
-
Participants: 2,981 patients with newly diagnosed glioblastoma enrolled in trials between 2008 and 2020.
-
Demographics: Median age 58 years; 62.3% male.
Bibliographic Data
-
Title: Proton Pump Inhibitor Use and Survival in Patients With Newly Diagnosed Glioblastoma
-
Authors: Le Rhun E, Sain D, Erridge SC, et al.
-
Journal: JAMA Network Open
-
Year: 2025
-
DOI: 10.1001/jamanetworkopen.2025.45578
This AI-generated analysis is for informational and research purposes only and is not a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of a qualified health provider with any questions you may have regarding a medical condition.
Original Article:
Full text pdf: Proton Pump Inhibitor Use and Survival in Patients with newly dx glioblastoma
CC-BY License Permissions
This is an open access article distributed under the terms of the CC-BY license, which permits unrestricted use, distribution, and reproduction in any medium. You are not required to obtain permission to reuse this article content, provided that you credit the author and journal.
