Comment:
This 15-year mortality data from ProtecT is a powerful addition to the 12-year patient-reported outcomes (PROs). underscoring the equivalence between the groups in terms of overall survival, but differences in functional outcomes.
We already knew from the PROs analysis that generic quality-of-life scores were similar across all three groups (Active Monitoring, Prostatectomy, Radiotherapy) by year 12, but the functional outcomes were drastically different. Radical treatment meant significantly worse urinary and sexual function compared to active monitoring, with radiotherapy also carrying a higher risk of fecal leakage.
Now, with this 15-year update we are still seeing no significant difference in overall or prostate cancer-specific mortality (P=0.53) across the three arms. It validates the central message: for the majority of men, radical treatment does not extend life, but it certainly impacts quality of life.
The real question for each patient is now clear – is the halving of metastases and the need for long-term ADT worth the cost of an 18-24% chance of significant urinary leakage (in the prostatectomy arm) or the fecal leakage risk with radiotherapy? With a 97% survival rate regardless of treatment we need to focus on managing the risk of progression (metastases) rather than mortality. Every clinician counseling a patient should be using these two landmark papers—the PROs and the survival data—to guide truly an0 informed decision.
Summary:
This 15-year follow-up from the ProtecT trial provides strong evidence that for men with localized, PSA-detected prostate cancer, there is no significant difference in prostate cancer-specific mortality between active monitoring, prostatectomy, and radiotherapy. Survival from prostate cancer was high (approximately 97%) across all three groups. However, radical treatments (prostatectomy or radiotherapy) halved the incidence of metastases, local progression, and the need for long-term androgen-deprivation therapy compared to active monitoring. Since the low mortality benefit from radical treatment does not outweigh the risk of side effects, clinicians must focus on helping men weigh the trade-offs between the benefits of reducing progression and the harms associated with treatment side effects (e.g., urinary, bowel, and sexual function).
Results in Context
Primary Outcome
The primary outcome was death from prostate cancer, as adjudicated by an independent cause-of-death committee.
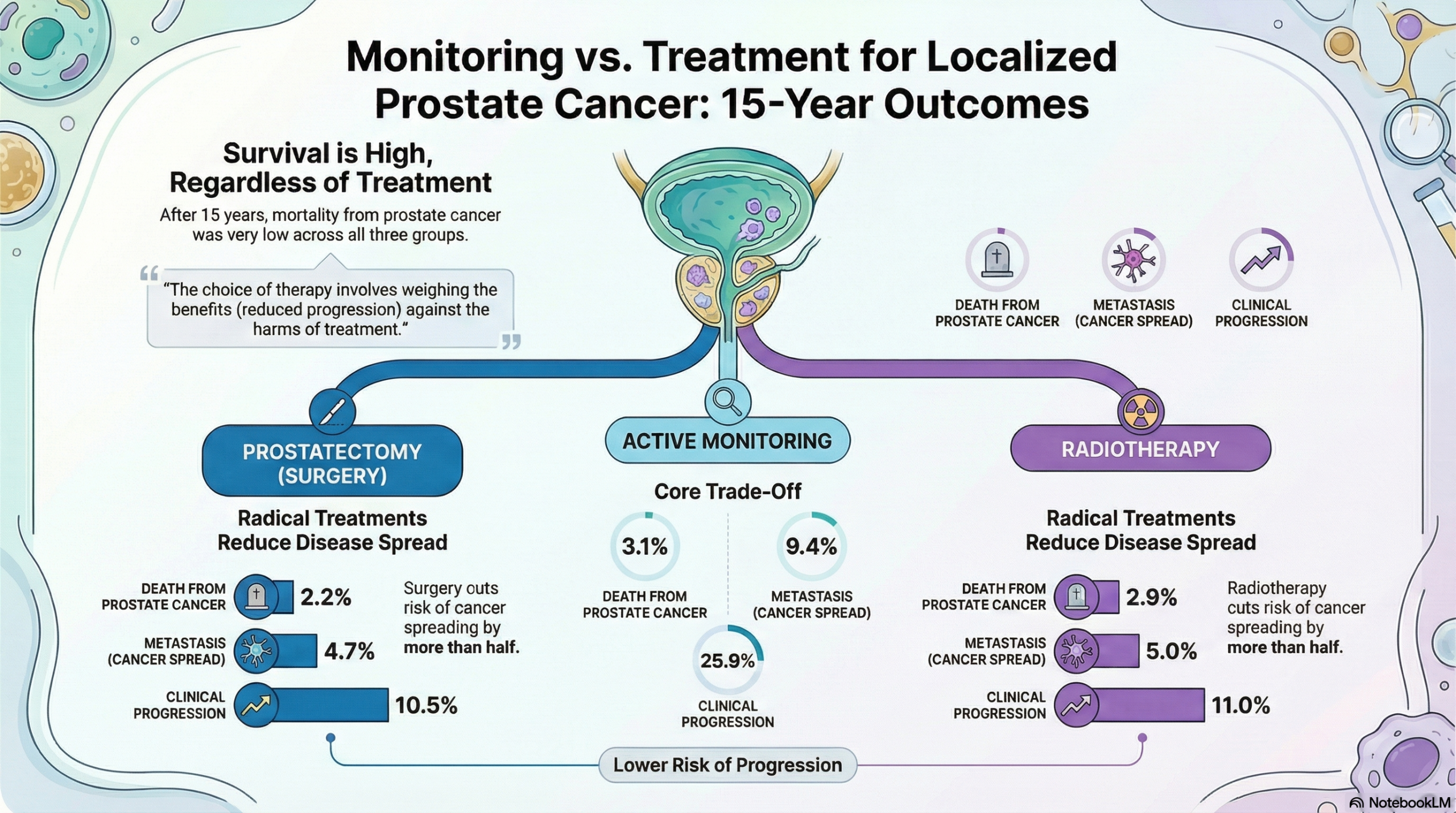
- After a median follow-up of 15 years, death from prostate cancer occurred in 45 men (2.7%) across all groups.
- The rates were similar in all three groups: 17 men (3.1%) in the active-monitoring group, 12 men (2.2%) in the prostatectomy group, and 16 men (2.9%) in the radiotherapy group.
- There was no significant difference in prostate cancer-specific mortality among the trial groups (P=0.53). Prostate cancer-specific survival was approximately 97% regardless of the treatment assignment.
Key Secondary & Specialized Outcomes
- Disease-Specific Survival Endpoints: The primary outcome of prostate cancer-specific mortality (death from the specific disease) was used. This differs from Overall Survival (OS) (death from any cause). Death from any cause occurred in 356 men (21.7%) with a similar distribution across the three groups, emphasizing that most deaths were not from prostate cancer.
- Metastatic Disease: Metastases developed in significantly fewer men in the radical treatment groups compared to active monitoring:
- Active Monitoring: 51 men (9.4%)
- Prostatectomy: 26 men (4.7%)
- Radiotherapy: 27 men (5.0%)
- Clinical Progression: The rate of clinical progression, a composite endpoint (including metastases, T3/T4 disease, long-term androgen-deprivation therapy initiation, and other adverse events), was also significantly lower in the radical treatment groups:
- Active Monitoring: 141 men (25.9%)
- Prostatectomy: 58 men (10.5%)
- Radiotherapy: 60 men (11.0%)
- Long-term Androgen-Deprivation Therapy (ADT): Initiated less often in the radical treatment groups:
- Active Monitoring: 69 men (12.7%)
- Prostatectomy: 40 men (7.2%)
- Radiotherapy: 42 men (7.7%)
- Harms and Adverse Events: The paper reports no differential effects on cancer-specific mortality were noted in relation to the baseline PSA level, tumor stage or grade, or risk-stratification score. Importantly, no new treatment complications were reported after the 10-year analysis. Patient-reported outcomes (PROs) regarding the full trade-offs between benefits and harms were described in a separate article.
Assertive Critical Appraisal
Risk of Bias (RoB 2 Framework)
Overall Risk of Bias: Low
The trial is a randomized controlled trial (RCT) with a high rate of patient follow-up (98%). The primary outcome, death from prostate cancer, was adjudicated by an independent cause-of-death committee. The 15-year analysis remained an intention-to-treat analysis. These factors significantly reduce the risk of bias in the primary outcome.
Subgroup Analyses
Subgroup analyses suggested a potential age-based difference in treatment effect that warrants caution.
- Men younger than 65 years: Active monitoring or prostatectomy had a lower risk of death from prostate cancer than radiotherapy.
- Men 65 years or older: Prostatectomy or radiotherapy had a lower risk of death from prostate cancer than active monitoring.
However, the authors state these are only a suggestion of an age effect and that they should be interpreted cautiously and warrant further exploration. Subgroup claims, especially those derived from interaction terms, are inherently unreliable unless the effect is large and pre-specified; therefore, this finding should be viewed with skepticism until confirmed by other trials.
Composite Endpoints
The secondary outcome of clinical progression is a composite endpoint composed of metastases, clinical T3 or T4 disease, initiation of long-term androgen-deprivation therapy, ureteric obstruction, rectal fistula, or urinary catheterization due to tumor growth. While the composite endpoint was significantly higher in the active-monitoring group (25.9%) compared to prostatectomy (10.5%) and radiotherapy (11.0%), the components are not of similar clinical importance. For example, local T3/T4 disease or a catheter due to tumor growth is less clinically significant than distant metastases. However, the treatment effect was consistent across major components, with active monitoring also showing a much higher rate of metastases (9.4% vs. ~5%) and ADT initiation (12.7% vs. ~7.5%).
Appraisal of Patient-Reported Outcomes (CONSORT-PRO)
Patient-reported outcomes (PROs), which are crucial for assessing the trade-offs between treatment benefits and harms, are described in a separate article. Therefore, a full appraisal of PRO methodology cannot be performed on this document, but their importance for clinical decision-making is correctly emphasized.
Reporting Quality Assessment (CONSORT)
The paper’s reporting quality is high. A participant flow diagram (Figure 1) is included, which clearly displays the number of men at each stage of the trial and follow-up, which is a key CONSORT requirement. The design is clearly a randomized trial, with detailed descriptions of the treatments and follow-up protocols.
Applicability
The findings are highly relevant to general clinical practice. The trial addresses the core controversy in localized prostate cancer management—whether to treat immediately or monitor. The cohort was population-based, consisting of men aged 50 to 69 years with PSA-detected, clinically localized disease. The treatments were standard conventional treatments (active monitoring, prostatectomy, radiotherapy). The generalizability of findings is considered a strength of the trial. The primary limitation on applicability is that the trial was conducted between 1999 and 2009, before the widespread use of modern diagnostic methods like multiparametric MRI and targeted biopsies. This means the contemporary “active surveillance” standard is more rigorous than the “active monitoring” protocol used in this trial.
Research Objective
The objective was to evaluate the effectiveness of active monitoring, prostatectomy, and radiotherapy on prostate cancer-specific and all-cause mortality, metastases, disease progression, and the initiation of long-term androgen-deprivation therapy after a median follow-up of 15 years in men with clinically localized prostate cancer.
Study Design
- Describe the trial design: This was a three-arm, parallel-group Randomized Controlled Trial (RCT) that randomly assigned men with localized prostate cancer to receive active monitoring (545 men), prostatectomy (553 men), or radiotherapy (545 men).
- Allocation and Blinding: The men were randomly assigned to a treatment group. Due to the nature of the interventions (surgery, radiation, or monitoring), it was an open-label trial (no blinding).
- Modern Trial Designs: The trial used a conventional, pragmatic RCT design.
Setting and Participants
- Setting and Locations: The trial was conducted in the United Kingdom between 1999 and 2009 at nine centers.
- Participants:
- 82,429 men between 50 and 69 years of age received a PSA test.
- 2664 men were diagnosed with localized prostate cancer and were eligible for treatment.
- 1643 men were enrolled in the trial and randomized to one of the three treatment arms.
- The median age at diagnosis was 62 years and the median PSA level was 4.6 ng/ml.
- At baseline, more than one third of the randomized men had intermediate or high-risk disease according to contemporary risk-stratification analysis.
Bibliographic Data
- Title: Fifteen-Year Outcomes after Monitoring, Surgery, or Radiotherapy for Prostate Cancer
- Authors: F.C. Hamdy, J.L. Donovan, J.A. Lane, C. Metcalfe, M. Davis, E.L. Turner, R.M. Martin, G.J. Young, E.I. Walsh, R.J. Bryant, P. Bollina, A. Doble, A. Doherty, D. Gillatt, V. Gnanapragasam, O. Hughes, R. Kockelbergh, H. Kynaston, A. Paul, E. Paez, P. Powell, D.J. Rosario, E. Rowe, M. Mason, J.W.F. Catto, T.J. Peters, J. Oxley, N.J. Williams, J. Staffurth, and D.E. Neal, for the ProtecT Study Group
- Journal: The New England Journal of Medicine
- Year: 2023
- DOI: 10.1056/NEJMoa2214122
This AI-generated analysis is for informational and research purposes only and is not a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of a qualified health provider with any questions you may have regarding a medical condition.
Original Article:
Full text: here
