Comment:
This analysis provides critical long-term data and most importantly context for localized prostate cancer treatment decision making. The most striking takeaway is the lack of difference in cancer-specific, or overall survival among the three groups (prostatectomy, radiotherapy, and active monitoring) over the 12-year follow-up.
Given that there is no difference in overall survival between the three, the focus must shift to properly informing patients of the differential adverse effects (AEs).
The functional harms are distinct, persistent, and significant:
-
Prostatectomy leads to the highest rates of long-term urinary leakage and the most severe, persistent decline in sexual function.
-
Radiotherapy carries the long-term burden of a persistent increase in fecal leakage.
-
Active Monitoring preserves functional outcomes best, deferring—and often avoiding entirely—the radical treatment-related AEs.
For newly diagnosed patients, this means the choice of treatment is predominantly a trade-off in quality-of-life harms, not a gamble on differences in survival. As clinicians, we need to have a detailed, balanced discussion where the long-term functional outcomes—urinary, sexual, and bowel—are presented as the primary decision drivers, not just secondary footnotes to the similar survival statistics. The patient’s personal tolerance for these specific, life-altering side effects should dictate the choice of management.
🔬Summary:
This analysis is based on the long-term follow-up of the ProtecT trial, a Randomized Controlled Trial (RCT) comparing treatments for localized prostate cancer.
Clinical Bottom Line
This long-term, randomized trial provides robust evidence that the three primary management strategies for localized prostate cancer—prostatectomy, radiotherapy, and active monitoring—continue to have distinct and persistent impacts on patient function over 12 years.
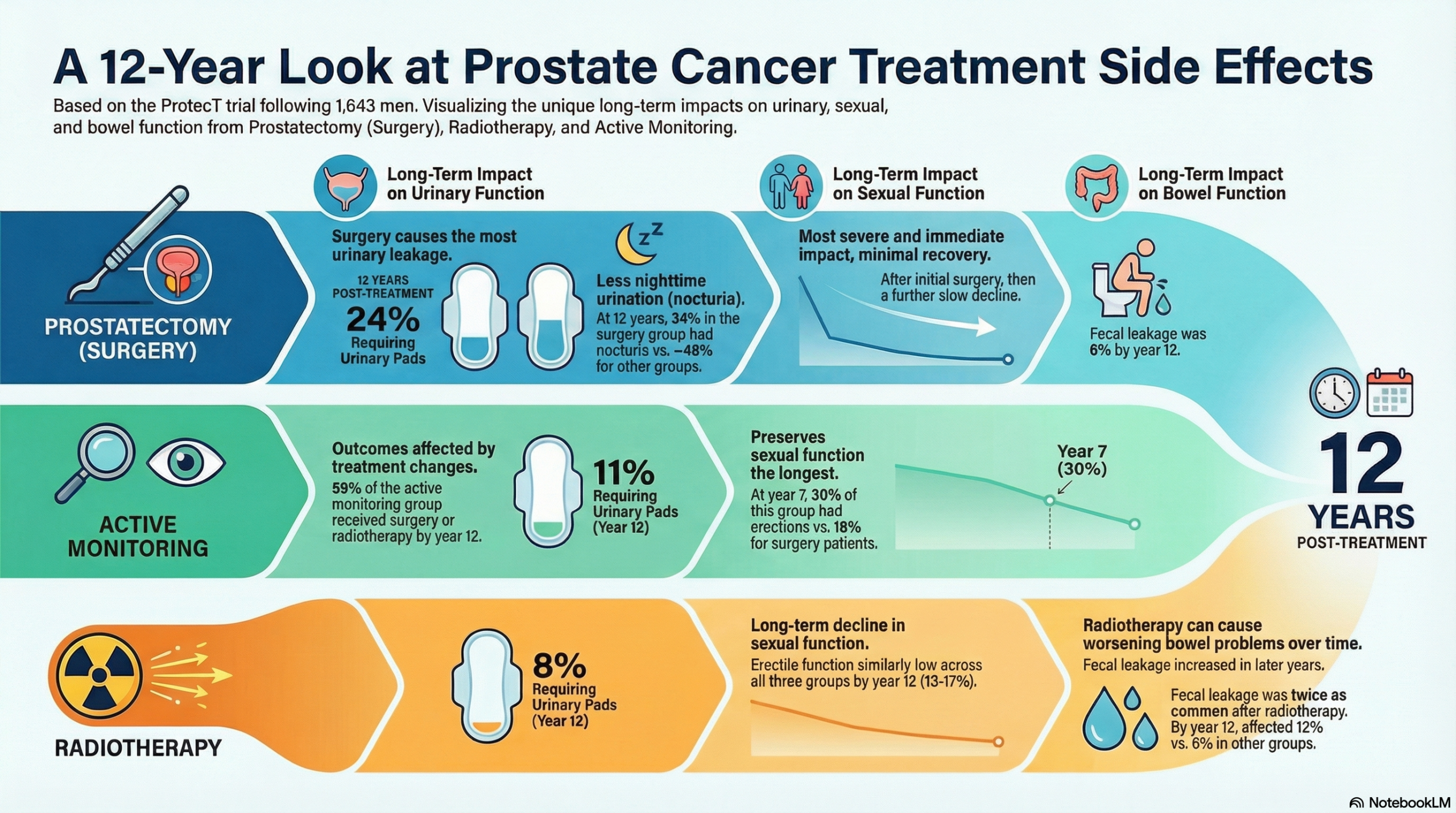
- Prostatectomy resulted in the worst long-term urinary leakage (requiring pads in 18-24% of patients by year 12) and the most severe, persistent impairment of sexual function.
- Radiotherapy led to a late-onset increase in fecal leakage (12% by year 12 vs. 6% in other groups) and a similar pattern of long-term sexual function decline as prostatectomy.
- Active monitoring had the best preservation of urinary and sexual function; functional declines observed were gradual and age-related, avoiding radical treatment effects unless the patient changed to a radical treatment.
- Generic quality-of-life scores (physical/mental health, anxiety, depression) were similar across all groups throughout the 7-to-12-year follow-up period.
These findings necessitate a detailed discussion with newly diagnosed patients about the trade-offs in long-term functional harms for each treatment.
Results in Context
Primary Outcome
The study focused on patient-reported outcomes (PROMs) over 7 to 12 years post-randomization, evaluating the functional and quality-of-life impacts of the three allocated treatments: prostatectomy, radiotherapy with neoadjuvant androgen deprivation, and active monitoring.
- Urinary Leakage (Requiring Pads): By year 12, 24% of the prostatectomy group used one or more pads per day, compared with 11% in the active monitoring group and 8% in the radiotherapy group. Urinary leakage was statistically significantly worse in the prostatectomy group.
- Nocturia (Voiding $\ge$ 2 times/night): At year 12, nocturia was more common in the active monitoring (47%) and radiotherapy (48%) groups, compared with the prostatectomy group (34%). This lower rate in the prostatectomy group may be due to relief of preexisting bladder outflow obstruction.
Key Secondary & Specialized Outcomes
Patient-Reported Outcomes (PROs): PROs were measured annually using validated tools, including the Expanded Prostate Index Composite (EPIC), International Consultation on Incontinence Questionnaire (ICIQ), and International Continence Society male Short Form (ICSmaleSF). The analysis used two-level mixed-effects linear and logistic models to assess outcomes over the 7-to-12-year period.
- Sexual Function (Erections Firm Enough for Intercourse):
- At 7 years, the percentage of men reporting potency was 18% for prostatectomy, 30% for active monitoring, and 27% for radiotherapy.
- By 12 years, all groups converged to similarly low levels (13% to 17%). The active monitoring group retained more sexual function for longer, while the prostatectomy group had the worst impact throughout.
- Bowel Function (Fecal Leakage): Fecal leakage (once per week or more) gradually increased in the radiotherapy group, affecting 12% by year 12, which was double the rate in the prostatectomy and active monitoring groups (6%).
- Generic Quality of Life: Generic quality-of-life scores (SF-12 Physical and Mental Health, HADS Anxiety/Depression) were similar across all randomized groups over the 7-to-12-year period.
Harms and Adverse Events
The study focused on long-term functional harms as the primary endpoints, which were measured using PROMs. The key harms were:
- Urinary Leakage: High and persistent rates following prostatectomy.
- Sexual Dysfunction: Severe and persistent impairment following prostatectomy, and a gradual decline over time in all groups.
- Fecal Leakage: Increased long-term risk associated with radiotherapy.
Assertive Critical Appraisal
Risk of Bias (RoB 2 Framework)
Overall Risk of Bias Judgment: Low
- Critique: The ProtecT trial is a high-quality randomized trial. The long-term follow-up and the use of patient-reported outcomes (PROMs) are critical strengths.
- Specific Strengths: The use of a randomized design ensures that the groups are balanced at baseline, enabling unbiased comparisons. The response rates exceeded 80% for most measures over the 12 years, which minimizes the risk of misleading results from missing data.
Subgroup Analyses
Subgroup analyses were performed for age ($<$ 65 vs. $\ge$ 65 years) and cancer risk stratification (low vs. intermediate/high).
- Critique: While subgroup findings should be viewed with skepticism, the observed differential effect on pad use for urinary leakage by age is plausible. Younger men in the active monitoring group were more likely to change management to prostatectomy (34% vs. 15% in older men), which likely explains the observed difference in urinary leakage within the active monitoring arm. There was no evidence of differential effects on other PROMs by cancer risk stratification.
Composite Endpoints
The study used several multi-item PROMs (e.g., EPIC, ICIQ, ICSmaleSF) that function as composite measures of quality of life or function.
- Critique: These tools are validated instruments designed to capture the complexity of functional domains. The authors addressed this by also presenting results for key individual items within the domains (e.g., pad use, erection firmness, fecal incontinence), allowing for a more granular, patient-centric interpretation.
Appraisal of Patient-Reported Outcomes (CONSORT-PRO)
- Critique: The PRO methodology in the ProtecT trial is a major strength.
- The PROMs were pre-specified.
- Response rates were sustained and very high (over 80% for most measures over 12 years), which is rare and mitigates the risk of bias from missing data.
- The statistical analysis (mixed-effects models) was appropriate for handling longitudinal, repeated measurements.
- The authors provided a clear rationale for presenting the data graphically without a single threshold for “clinical relevance,” correctly deferring judgment to the patient and clinician to consider individual values and priorities.
Reporting Quality Assessment (CONSORT)
- Critique: The reporting quality is high. While the paper itself does not include the participant flow diagram, it directs the reader to external sources where the study flowchart and clinical outcomes are detailed (Hamdy et al.). The design is well-described, including the randomization details (stratification/minimization variables).
Applicability
The findings are highly applicable to general clinical practice in prostate cancer management.
- The study included a population-based cohort of men with clinically localized prostate cancer (low- to intermediate-risk), which is directly relevant to most newly diagnosed patients.
- The results provide the most robust long-term data on the patient experience (PROMs) for the three main management strategies, which is essential for informed, shared decision-making given the high likelihood of long-term survival.
Research Objective and Study Design
Research Objective
The objective was to evaluate the long-term patient-reported outcomes of 1643 randomly assigned participants in the ProtecT trial, specifically focusing on the functional and quality-of-life impacts of prostatectomy, radiotherapy with neoadjuvant androgen deprivation, and active monitoring from 7 to 12 years after randomization.
Study Design
- Design: Parallel-group, randomized, phase 3 trial (long-term follow-up analysis).
- Allocation: 1643 participants with clinically localized prostate cancer were randomly assigned between 2001 and 2009:
- 545 to Active Monitoring (surveillance with PSA and clinical review).
- 553 to Radical Prostatectomy (mostly open retropubic).
- 545 to Radiotherapy (external beam with neoadjuvant androgen deprivation).
- Blinding: This was an unblinded trial given the nature of the interventions (surgery, radiation, active monitoring).
- Follow-up: Patient-reported outcome measures (PROMs) were completed at 6 months and then annually, with this analysis focusing on the 7-to-12-year outcomes.
Setting and Participants
- Setting: The study was the U.K. National Institute for Health and Care Research-supported ProtecT trial.
- Participants: 1643 men diagnosed with clinically localized prostate cancer following population-based PSA testing and 10-core biopsy. The cohort included at least one-third with intermediate-risk prostate cancer. The participants were recruited between 2001 and 2009.
Bibliographic Data
- Title: Patient-Reported Outcomes 12 Years after Localized Prostate Cancer Treatment
- Authors: Jenny L. Donovan, Ph.D., F.Med.Sci., et al.
- Journal: NEJM Evidence
- Year: 2023
- DOI: 10.1056/EVIDoa2300018
This AI-generated analysis is for informational and research purposes only and is not a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of a qualified health provider with any questions you may have regarding a medical condition.
Original Article:
Full text: here.
