💬Comment:
This is a pilot study and has a lot of weaknesses in terms of study design. However it also shows a median actual survival time 40% longer than predicted—17 months versus 12 months—in this highly selected group of end-stage patients.
The authors rightly point out the major methodological flaw of a retrospective, non-randomized, and uncontrolled design, and the consistency of the survival gain (76% of patients survived longer than predicted) is compelling. The findings suggest that the combination of Coenzyme Q10 and the other antioxidants may be doing something genuinely beneficial, and with an incredible safety profile, it’s something I generally recommend.
Summary:
Clinical Bottom Line
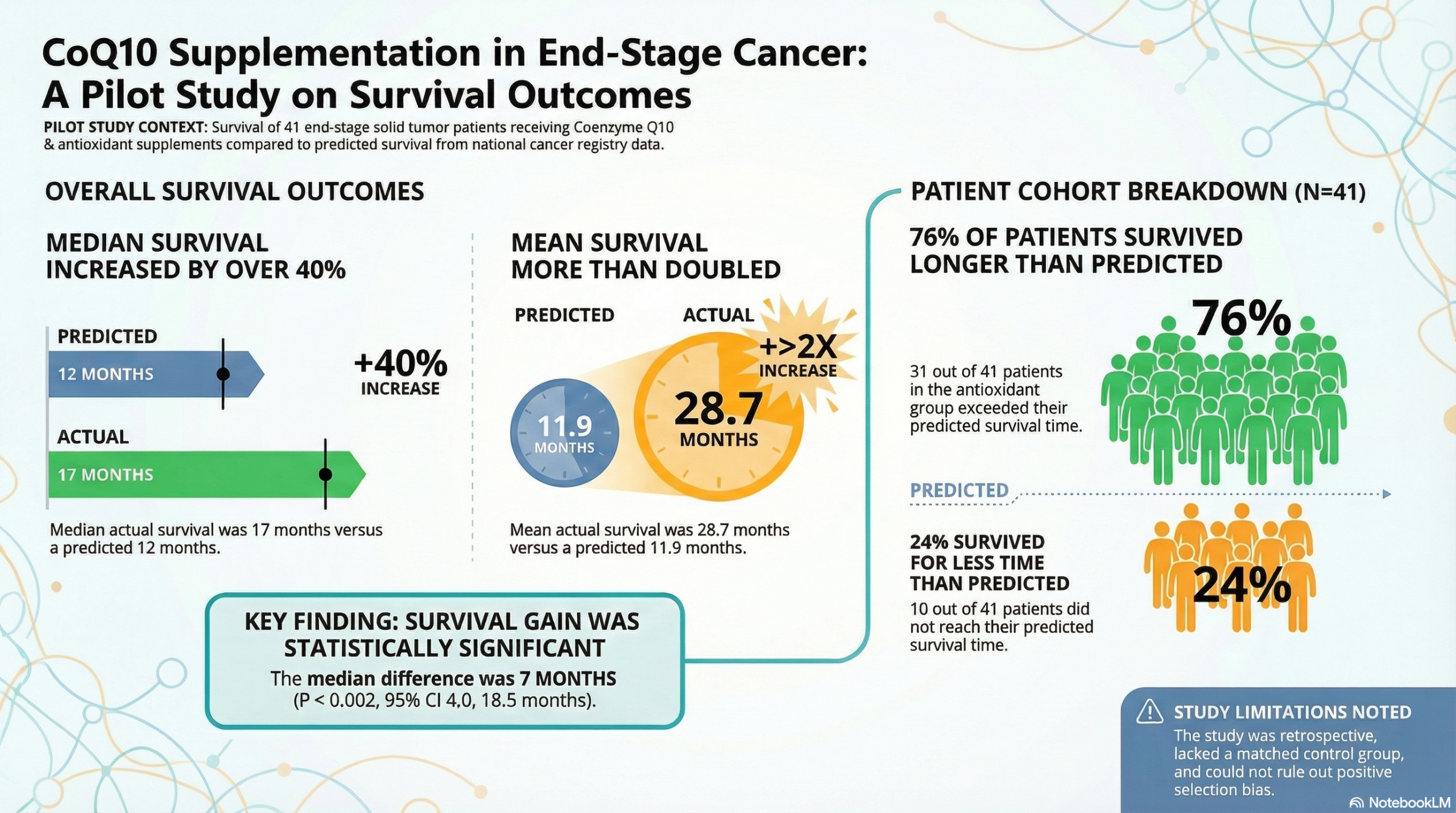
This observational pilot study suggests a strong association between the use of Coenzyme $Q_{10}$ and other antioxidant supplements and prolonged survival in a small, select group of patients with end-stage cancer. The median actual survival was $40%$ longer than the median predicted survival (17 months versus 12 months). Furthermore, the survival benefit was most pronounced in the subgroup of patients who began the antioxidant treatment earlier (within 1.5 months of incurable diagnosis).
However, due to the study’s retrospective, non-randomized, and uncontrolled design, it is not possible to conclude a causal link between the supplements and the survival gain. The results are highly susceptible to selection bias and unmeasured confounding variables, meaning the observed effect could be due to differences in the patients who chose and tolerated the treatment rather than the antioxidants themselves. This study serves as a strong signal to prioritize larger, controlled clinical trials.
Main Results
The study evaluated 41 consecutive patients with end-stage cancer who received the antioxidant supplement regimen.
- Median Survival: The median actual survival was 17 months (range 1–120 months), which was $> 40%$ longer than the median predicted survival of 12 months (range 3–29 months).
- The median difference (gain) in survival was 7 months, which was statistically significant ($P < 0.002$).
- Mean Survival: The mean actual survival was 28.7 months versus 11.9 months for the mean predicted survival.
- Proportion of Patients: 31 patients ($\mathbf{76%}$) survived for longer than predicted, while 10 patients ($24%$) survived for less time than predicted.
- Timing of Treatment: The survival gain appeared to be greater in patients who started treatment earlier.
- Patients ($\mathbf{n=20}$) who started antioxidant treatment within 1.5 months of diagnosis had a median actual survival in excess of the predicted time of 7 months.
- Patients ($\mathbf{n=21}$) who started treatment $> 1.5$ months after diagnosis had a median actual survival in excess of the predicted time of 1.5 months.
Intervention
Patients received a combination of supplements daily, including:
- Coenzyme $Q_{10}$ ($Q_{10}$): 300 mg
- Vitamin C: 5.7 mg
- Selenium (as selenomethionine): 487 $\mu$g
- Folic acid: 5 mg
- $\beta$-Carotene: 76 mg (patients with lung cancer did not receive $\beta$-carotene for safety reasons)
- $\alpha$-Tocopherol: 1.625 mg
- Vitamin A: 25,000 IU
Harms and Adverse Events
Treatments were very well tolerated with few adverse effects. Side effects were minor and rare, mainly consisting of difficulties swallowing the many tablets and an aversion to the odour of the tablets, especially as the patients’ general physical condition deteriorated. No other physical side effects were noted. Investigators reported that a large majority of patients experienced an impressive improvement in general well-being, although this was not formally measured.
Limitations & Bias (STROBE Framework)
This is a long-term observational pilot study and is highly susceptible to methodological limitations inherent to this design.
- No Matched Control Group: The most critical limitation is the lack of a contemporaneous, matched control group. The comparison is made against a predicted survival time calculated from historical Kaplan-Meier curves from the National Danish Cancer Registry, which only provides an estimated, not a true, control.
- Selection Bias (Major Flaw): There is a high potential for positive selection bias.
- Patients had to survive and continue treatment for $\ge 2$ months to be included in the evaluation, which excluded 29 extremely ill patients who died quickly. This immediately biases the included cohort toward those with better baseline prognoses who were healthy enough to tolerate the regimen, inflate the observed survival.
- Patients were self-selected or offered treatment in a Danish private practice setting. Patients who seek out and can afford alternative or supplementary therapy may have better socioeconomic status, access to care, or a more positive mindset, which are all confounding factors.
- Lack of Blinding: The study was open-label (unblinded), which is unavoidable in this design, but means the investigators’ strong impression of “impressive improvement of their general well-being” is highly subject to investigator and patient bias (a placebo effect).
- Lack of Ethical Approval: The study protocol did not undergo any ethical approval, as legal ethics review committees did not exist in Denmark until 1992. This is a historical note that would not meet modern research standards.
Reporting Quality Assessment (STROBE)
The study provides a detailed description of the patient population and outcomes. However, as an observational study, its ability to address confounding is its primary flaw.
- Confounding: The authors attempted to account for confounding by using prognostic models (Kaplan-Meier curves) specific to the type, stage, age, and sex of cancer where possible. However, the predicted survival estimates were often derived from crude or historical data that did not account for all modern prognostic factors (e.g., oestrogen receptor status for breast cancer, or recent chemotherapy protocols for colorectal cancer). This failure to adequately address all potential confounders is a major flaw that reduces the confidence in the magnitude of the survival gain reported.
Applicability
The findings suggest a potential benefit of this antioxidant combination in advanced cancer. The treatments were well-tolerated, which is a practical consideration for end-stage patients. However, the patient population was highly heterogeneous (multiple primary cancer types and stages) and highly select, limiting the generalizability of the exact magnitude of benefit to a broader, unselected clinical population.
Research Objective
The objective of this pilot study was to evaluate the survival of patients with end-stage cancer who received supplements of coenzyme $Q_{10}$ and a mixture of other antioxidants as a supplement to their usual cancer therapy.
Study Design
This was a long-term, observational pilot study conducted in a Danish private practice setting.
- Design: Observational, retrospective evaluation of consecutive patients.
- Inclusion Period: Patients were included for a period of 9 years (January 1990 and April 1999) and followed up for over 15 years.
- Treatment: Patients were offered the antioxidant regimen as a supplement to their usual cancer therapy.
- Primary Comparison: Actual survival was compared to the predicted survival time, which was estimated mainly using Kaplan-Meier curves from the National Danish Cancer Registry and previously published prognostic data.
Setting and Participants
- Setting: A Danish private practice.
- Participants:
- Total Evaluated: 103 patients visited the clinic and were evaluated.
- Included in Study: 41 patients with end-stage cancer (solid tumors with distant metastases or inoperable) were included.
- Exclusions: 29 patients were excluded because they survived and continued treatment for $< 2$ months. Others were excluded for having non-solid tumors or only regional/local cancer.
- Primary Cancer Sites: Included breast, brain, lungs, kidneys, pancreas, oesophagus, stomach, colon, prostate, ovaries, and skin.
Bibliographic Data:
Title: Improved Survival in Patients with End-stage Cancer Treated with Coenzyme $Q_{10}$ and Other Antioxidants: a Pilot Study
Authors: N Hertz and RE Lister
Journal: The Journal of International Medical Research
Year: 2009
DOI: 10.1177/147323000903700634
Mandatory Disclaimer: This AI-generated analysis is for informational and research purposes only and is not a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of a qualified health provider with any questions you may have regarding a medical condition.
Original Article:
Full text: Available here.
