Comment:
The ECLIPSE trial presents a conflict between the accessibility of a blood-based “liquid biopsy” and the structural reality that it fails to detect the early lesions that make colorectal screening a preventive success.
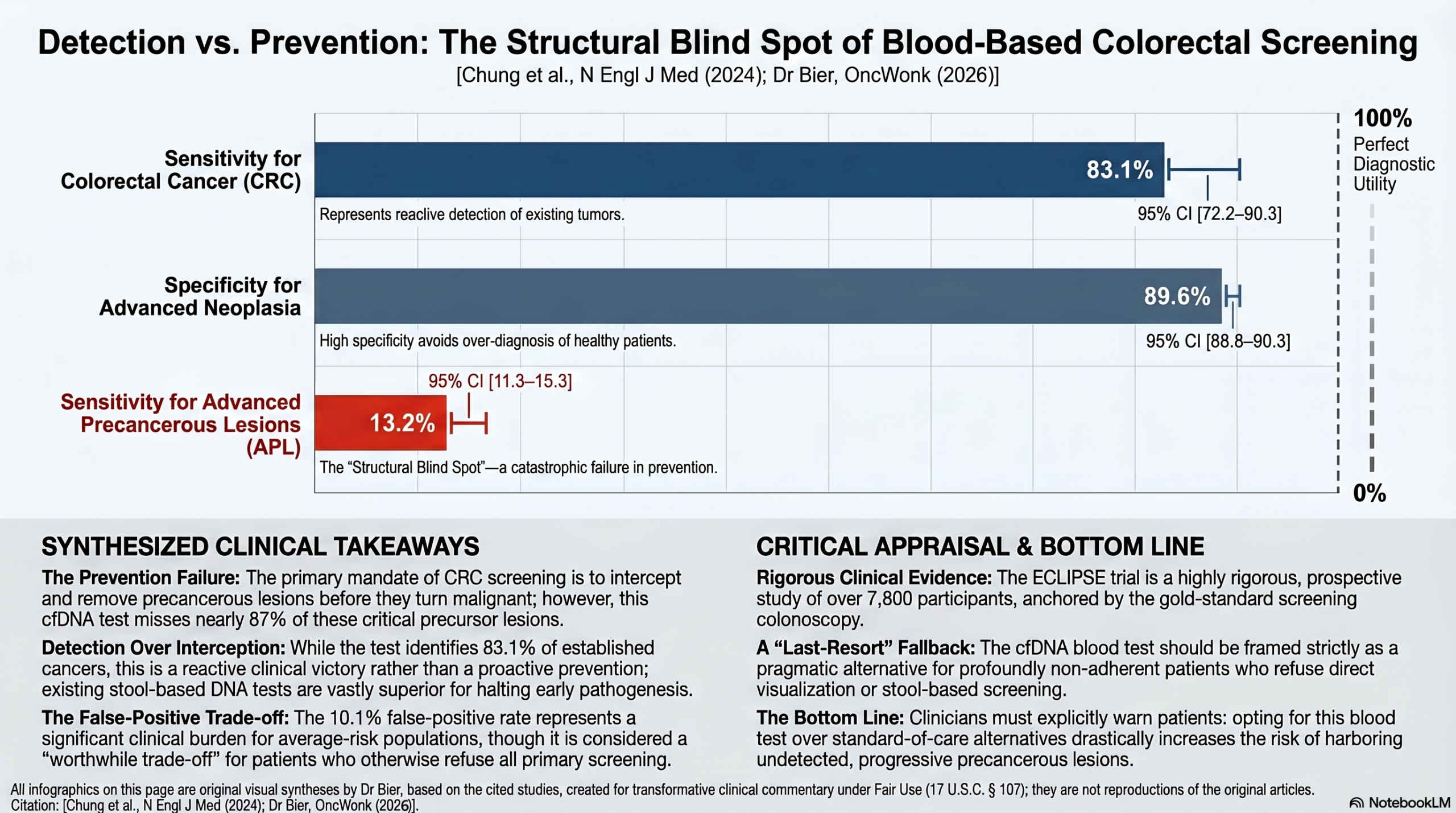
I’m excited about the possibilities of cfDNA technology, but we have to be transparent about its limitations. While an 83.1% sensitivity for established cancer is a functional net for those who refuse traditional screening, the test fails at cancer prevention. Its sensitivity for advanced precancerous lesions (APLs) is a dismal 13.2%. By comparison, standard stool-based tests like FIT (23%) or multitarget DNA (43%) are significantly more effective at identifying the precursors we actually want to remove. To offer this test without highlighting this 87% miss rate for precancer is offering a false sense of security.
We should also be aware of the relatively high level of false positive results. A specificity of 89.6% means the test carries an approximately 10% false-positive rate. Of course, if this is used in a population that would normally refuse colonoscopy screening, that is likely a worthwhile trade-off.
I am recommending this test only as a secondary tool for the roughly one-third of the population that is currently non-adherent to any screening. It is a “cancer detector,” not a “prevention shield.” It is not a one-to-one replacement for colonoscopy or even stool-based testing. Its best use is for those who would otherwise remain completely unscreened—provided they understand that while the test may find a tumor, it is mathematically likely to miss the window to prevent one.
The Wonk Debate – Audio Critique & Clinical Commentary:
Summary:
Clinical Bottom Line
This large-scale diagnostic accuracy study (ECLIPSE) demonstrates that a cell-free DNA (cfDNA) blood test (Shield) has an acceptable sensitivity for detecting colorectal cancer (83%) and high specificity (90%) in an average-risk population. However, its clinical utility for cancer prevention is significantly limited by its poor sensitivity for advanced precancerous lesions (13%), which is notably lower than existing stool-based options. While the test may improve screening adherence due to its convenience, clinicians must clearly communicate that it is less effective at identifying the lesions that, if removed, prevent cancer from developing.
Results in Context
Main Results (Diagnostic Accuracy Metrics)
- Sensitivity for Colorectal Cancer (CRC): 83.1% (95% CI, 72.2 to 90.3).
- False Negative Rate for CRC: 16.9% (11 of 65 cancers were missed).
- Specificity for Advanced Neoplasia: 89.6% (95% CI, 88.8 to 90.3).
- Specificity for Negative Colonoscopy: 89.9% (95% CI, 89.0 to 90.7).
- False Positive Rate: 10.1% (among participants with no neoplasia on colonoscopy).
- Sensitivity for Advanced Precancerous Lesions (APL): 13.2% (95% CI, 11.3 to 15.3).
Definitions & Context This assay is a diagnostic (screening) biomarker intended to identify asymptomatic individuals who require a diagnostic colonoscopy.
- Sensitivity: The ability of the test to correctly identify those with the disease.
- Specificity: The ability of the test to correctly identify those without the disease.
- Positive Predictive Value (PPV): In a theoretical population of 100,000, the PPV for CRC was 3.2%, meaning roughly 3 out of 100 “abnormal” results actually represent a cancer.
Biomarker Technology The test (Shield, Guardant Health) utilizes a multimodal approach to analyze cell-free DNA (cfDNA)—small fragments of DNA released into the bloodstream. The panel interrogates genomic alterations, aberrant methylation status, and fragmentomic patterns to provide a binary “abnormal” or “normal” signal.
Assertive Critical Appraisal
Appraisal of Biomarker Claim (Screening/Diagnostic) The authors position this test as a tool to bridge the screening gap, as one-third of the eligible population remains unscreened. While the study design is a robust, prospective multicenter trial (265 sites) that appropriately used the gold standard (colonoscopy) for all participants, a major clinical caveat exists.
- Detection vs. Prevention: The primary strength of colorectal screening is the detection and removal of advanced precancerous lesions (APLs). The sensitivity for APLs here (13.2%) is substantially lower than the multitarget stool DNA test (43.4%) or even a single FIT (23.3%). This means the blood test is much more likely to miss the window for cancer prevention.
Reporting Quality Assessment (REMARK)
- Assay Methodology: The assay thresholds and panel were locked prior to analysis, which is a critical strength for validating a diagnostic tool.
- Population Specification: The study clearly defined the average-risk screening population, including the newly recommended 45–49 age group.
- Cut-point determination: The statistical methods for determining a “positive” test were pre-specified and aligned with FDA-approved criteria for other screening tests.
Applicability The test is highly applicable to general clinical practice as it can be performed during a routine blood draw, potentially overcoming barriers like the unpleasantness of stool collection or the logistics of colonoscopy. However, its effectiveness as a “screening program” depends on two levels of adherence: adherence to the blood test and, crucially, adherence to follow-up colonoscopy for those with a positive result.
Research Objective
The study aimed to evaluate the performance of a cfDNA blood-based test (Shield) for the detection of colorectal cancer in an average-risk population undergoing routine screening colonoscopy.
Study Design
This was a prospective, observational, multicenter clinical validation study (ECLIPSE).
- Cohort: 22,877 participants were enrolled; 7,861 were evaluable for the final analysis after down-sampling the negative-colonoscopy group to meet statistical power requirements.
- Reference Standard: Every participant underwent a screening colonoscopy, with histopathological review of resected lesions.
Setting and Participants
- Settings: 265 sites in the U.S., including academic and community health centers across 38 states.
- Participants: Average-risk adults aged 45 to 84 years. The cohort was racially and ethnically diverse, closely mirroring 2020 U.S. Census data (78.5% White, 11.8% Black, 7.1% Asian, 13.3% Hispanic).
Bibliographic Data
- Title: A Cell-free DNA Blood-Based Test for Colorectal Cancer Screening
- Authors: Daniel C. Chung, M.D., Darrell M. Gray II, M.D., M.P.H., Harminder Singh, M.D., et al.
- Journal: The New England Journal of Medicine
- Year: 2024
- DOI: 10.1056/NEJMoa2304714
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
