Comment:
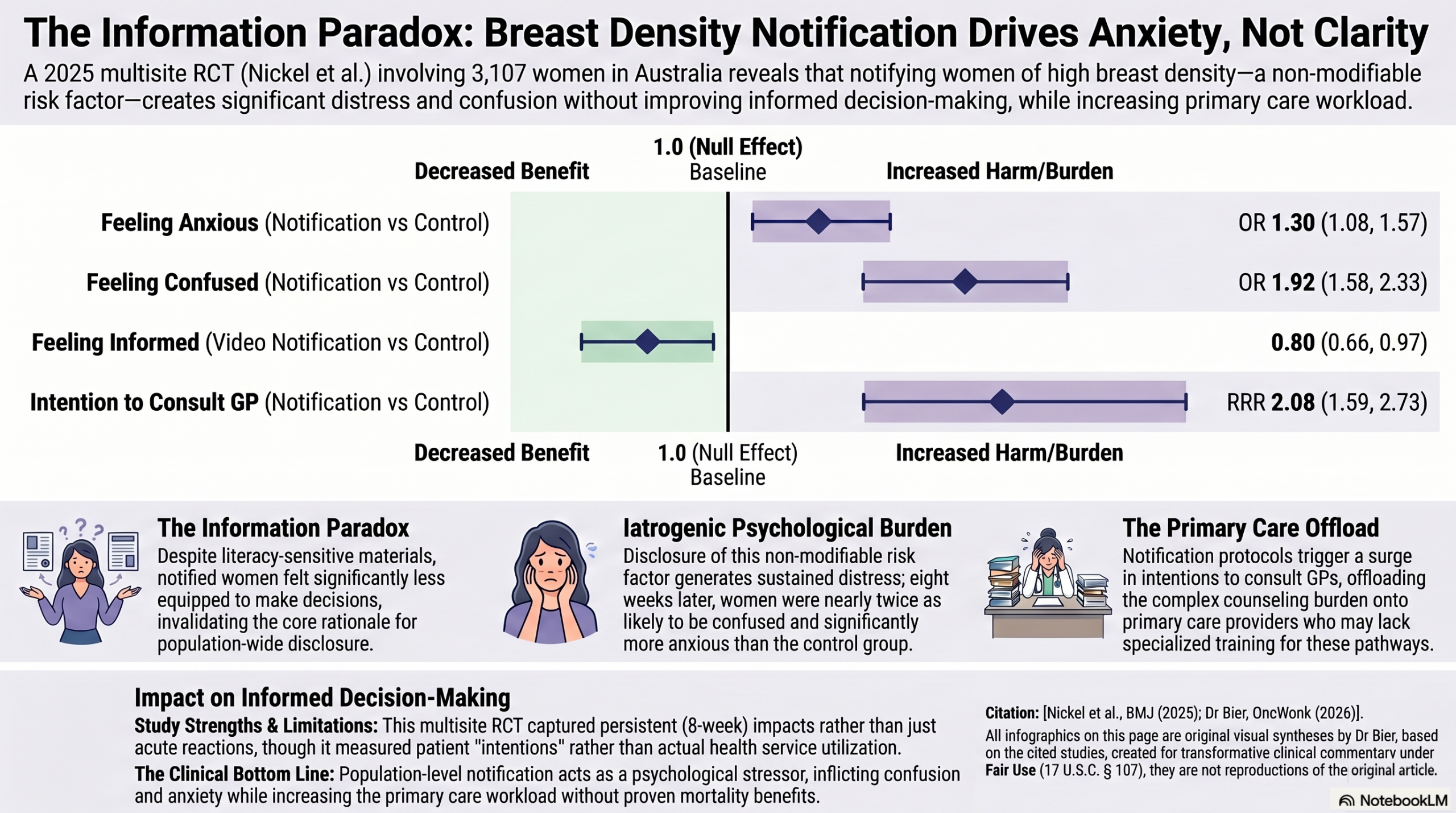
This is a difficult one for me, as I am a strong proponent of Informed Medicine. Patients need the information so they can balance the risks and benefits. And yet, the findings from this 2025 RCT demonstrate a structural failure in the notification paradigm. Providing more data resulted in patients not feeling more informed, even trending towards less informed although not significant under stricter statistical comparison criteria. Women who received density notifications were significantly more likely to report feeling anxious and nearly twice as likely to feel confused and had no better odds of feeling informed to make decisions about their breast health. The data shows that after 8 weeks, these women were still more anxious and confused than those who received standard care.
While notification significantly increased the intention to seek advice from general practitioners (RRR 2.08), it barely moved the needle on actual supplemental screening intent—79–81% of women still had no intention to pursue it. Notified women were also significantly more likely to report that they would rely on their GP to decide on supplemental screening (RRR 2.61), effectively offloading the burden of a complex decision onto a general practitioner who very likely lacks the specialized knowledge to manage it.
The Wonk Debate – Audio Critique & Clinical Commentary:
Summary:
Clinical Bottom Line:
This large-scale randomized trial provides clear evidence that notifying women of their high breast density increases psychological harms, specifically anxiety and confusion, without improving how informed they feel about their breast health. While the notification significantly increased the intention to seek further advice from general practitioners (GPs), it did not lead to a widespread desire for supplemental screening. Clinicians should be aware that population-level notification may impose a substantial counseling burden on primary care without achieving the intended goal of better-informed patient decision-making.
Results in Context
- Psychological Outcomes (Primary Harm): Women notified of dense breasts reported significantly higher levels of:
- Anxiety: Both notification methods increased anxiety compared to standard care (Written info: OR 1.30, 95% CI 1.08–1.57; Video link: OR 1.28, 95% CI 1.07–1.54).
- Confusion: Notified women were nearly twice as likely to feel confused (Written info: OR 1.92, 95% CI 1.58–2.33; Video link: OR 1.76, 95% CI 1.46–2.13).
- Perceived Knowledge: Critically, notified women did not feel more informed than those who received no notification (Video link: OR 0.80, 95% CI 0.66–0.97).
- Health Service Use Intentions: Notification led to a marked increase in the intention to rely on primary care for guidance:
- GP Consultation: Notified women were significantly more likely to plan to talk to their GP about their results (Written info: RRR 2.08; Video link: RRR 1.71).
- Supplemental Screening Advice: There was a strong shift toward relying on GPs to decide on extra screening (Written info: RRR 2.61; Video link: RRR 2.29).
- Screening Intent: Despite the notification, 79–81% of women still did not intend to have supplemental screening.
Harms and Adverse Events
The “harms” in this trial were defined as the unintended psychological consequences of medical notification.
- Heightened Anxiety and Confusion: The trial confirms that breast density notification triggers psychological distress that is measurable and sustained at 8 weeks post-screening. Notified women were significantly more likely to report feeling anxious (OR 1.30) and confused (OR 1.92) compared to the control group. This indicates that the notification creates a lingering state of unease rather than a brief, transitory reaction.
- Failure to Inform: A key harm is the “information paradox”—women were given more data but felt less informed, suggesting the “literacy-sensitive” materials did not overcome the inherent complexity of the topic.
- Health System Burden: The trial highlights a “consultation burden” on GPs, who must now navigate these complex conversations without clear clinical evidence that supplemental screening improves outcomes for this specific population.
Assertive Critical Appraisal
- Risk of Bias (RoB 2): Low. The trial utilized robust computer-generated randomization and maintained concealment until the intervention stage.
- Patient-Reported Outcomes (CONSORT-PRO): The use of psychological scales at 8 weeks is appropriate for capturing the impact of notification. However, the study focuses on intentions rather than actual health service use, which may not perfectly predict real-world GP visit rates.
- Applicability: These findings are highly relevant to current international debates where breast density notification is being legislated (e.g., in the US). It demonstrates that “health literacy sensitive” materials may still fail to mitigate the confusion caused by notifying women of a non-modifiable risk factor.
- Reporting Quality Assessment (CONSORT): The paper provides clear details on participant flow (randomizing 3,107 women) and methodology, aligning with standard reporting guidelines.
Research Objective
To investigate the effect of notifying women of their dense breasts on their psychosocial outcomes and health service use intentions compared to standard care.
Study Design
Multisite, parallel-arm, randomized controlled trial with three groups: standard care (no notification), notification + written information, and notification + video link.
Setting and Participants
3,107 women aged ≥40 years undergoing population-based breast screening in Queensland, Australia, who were identified as having dense breasts (BI-RADS C or D).
Bibliographic Data
- Title: Impact of population based breast density notification: multisite parallel arm randomised controlled trial in BreastScreen
- Authors: Brooke Nickel, Nick Ormiston-Smith, Erin Cvejic, et al.
- Journal: BMJ
- Year: 2025
- DOI: 10.1136/bmj-2024-083649
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
