Comment:
The central justification for aspirin use in primary prevention has long relied on the “Legacy Effect”—the theory that years of early exposure act as a biological investment, paying dividends in reduced cancer risk decades later. This follow-up to the ASPREE trial effectively demolishes that hypothesis for older adults. We are witnessing a stark collision between the conventional wisdom derived from middle-aged cohorts (like the Women’s Health Study) and the biological reality of the geriatric population: in adults over 70, aspirin provides no delayed protection and instead acts as an active promoter of cancer mortality.
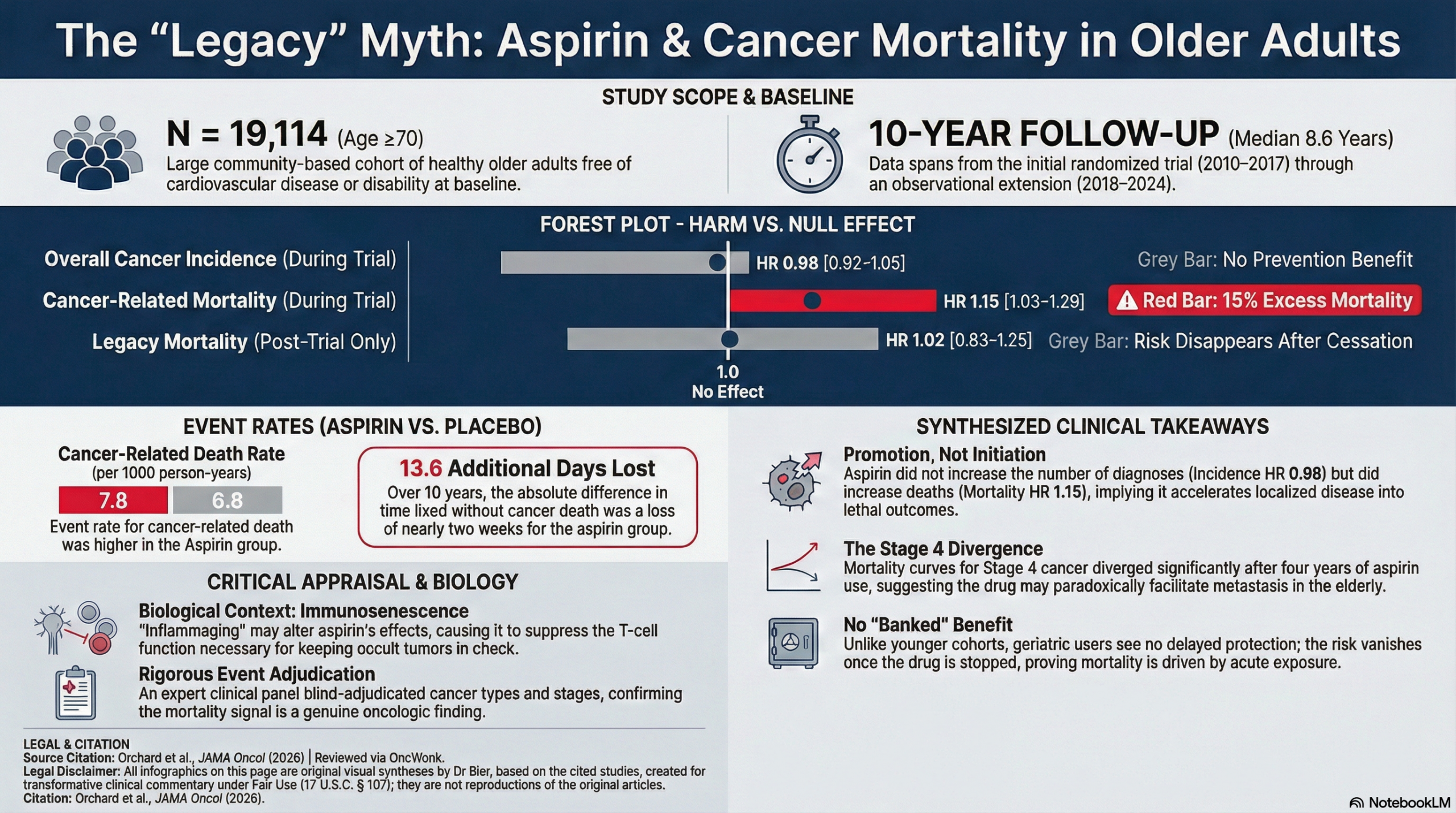
Applying the “Guilty Until Proven Innocent” paradigm, aspirin has failed to demonstrate efficacy and has decisively proven its guilt regarding hard clinical endpoints. Over a median of 8.6 years, aspirin did not reduce overall cancer incidence (HR = 0.98; 95% CI, 0.92-1.05). Far more concerning is the hierarchy of harm: while incidence remained neutral, cancer-related mortality was significantly elevated by 15% (HR = 1.15; 95% CI, 1.03-1.29). This discrepancy implies that while aspirin may not initiate the cancer, it likely accelerates its progression, converting survivable disease into lethal metastatic outcomes—a mechanism supported by the observation that Stage 4 cancer deaths diverged significantly after four years.
The most critical analytical insight lies in the “Legacy” analysis itself. When the specific impact of the post-trial period was isolated, the elevated mortality risk vanished (HR = 1.02; 95% CI, 0.83-1.25). While the authors frame this lack of persisting harm as a relief, it confirms a disturbing pharmacokinetic reality: the increased death rate was driven by acute exposure. The drug was actively suppressing immunosurveillance or facilitating metastasis while in the system, and that toxicity only ceased when the intervention stopped. There is no “banked benefit” to be gained, only immediate risk.
The Wonk Debate – Audio Critique & Clinical Commentary:
Summary:
Clinical Bottom Line
For healthy older adults (≥70 years) without existing cardiovascular disease, initiating a daily 100-mg aspirin regimen provides no protective benefit against cancer incidence over a 10-year period. Critically, this strategy is associated with a 15% increase in cancer-related mortality. These findings reinforce that initiating multiyear aspirin for cancer prevention is not recommended in the elderly. Unlike studies in middle-aged populations, this trial shows no “legacy effect” where benefits emerge years after treatment cessation; instead, the early increased mortality risk appears to be a persistent residuum of the initial trial phase.
Results in Context
Main Results: Cancer Incidence and Mortality
- Overall Cancer Incidence: Over a median follow-up of 8.6 years, low-dose aspirin (LDA) was not associated with a reduction in the first occurrence of cancer (HR = 0.98; 95% CI, 0.92-1.05).
- Cancer-Related Mortality: LDA was associated with a statistically significant increase in cancer deaths (HR = 1.15; 95% CI, 1.03-1.29).
- Definitions: A hazard ratio (HR) of 1.15 means there was a 15% higher hazard (rate) of cancer death in the aspirin group compared to the placebo group during the study period.
- Event Rates: The rate of cancer death was 7.8 per 1000 person-years for the aspirin group versus 6.8 per 1000 person-years for the placebo group.
Key Findings by Stage and Type
- Stage 4 Cancer: The increased mortality risk is potentially driven by a higher incidence of Stage 4 (metastatic) cancers observed in the aspirin group during the randomized phase.
- Colorectal Cancer (CRC): Despite prior evidence in younger populations, there was no reduction in CRC incidence (HR = 1.01; 95% CI, 0.84-1.21).
- Melanoma: A possible long-term protective effect was noted for melanoma incidence (HR = 0.77; 95% CI, 0.62-0.94), though authors caution this may be a chance finding.
- Restricted Mean Time Lost (RMTL): The absolute difference in time lived without cancer death was an additional 13.6 days lost in the aspirin group over 10 years.
Legacy Effects (Post-Trial Observation)
- Incidence & Mortality: Among participants who survived the RCT without cancer, original aspirin assignment did not affect subsequent cancer incidence (HR = 0.91; 95% CI, 0.82-1.01) or mortality (HR = 1.02; 95% CI, 0.83-1.25).
- Metastatic Benefit: A lower incidence of new metastatic cancer was observed in the post-RCT phase for those originally on aspirin (HR = 0.77; 95% CI, 0.60-0.97), but this did not translate to improved survival.
Assertive Critical Appraisal
Limitations & Bias (STROBE Framework)
- Biological Context: The authors hypothesize that “immunosenescence” (age-related immune decline) or “inflammaging” (chronic inflammation) in older adults might cause aspirin to paradoxically blunt the body’s natural immunosurveillance against cancer.
- Adherence and Unblinding: While the RCT phase was double-blinded, the extension (ASPREE-XT) was observational. However, post-trial aspirin use (including open-label use) was balanced between groups, minimizing this as a source of bias.
- Follow-up Duration: While 8.6 years is substantial, some cancer types (like CRC) may require 10–20 years for a preventive effect to fully manifest.
- Multiple Testing: Multiple subgroups and cancer types were analyzed without statistical correction for multiple comparisons, meaning findings like the melanoma benefit or brain cancer risk (HR = 1.96) should be interpreted with caution.
Reporting Quality Assessment (STROBE)
- Confounding: The authors explicitly described efforts to address confounding, confirming that baseline characteristics remained well-balanced even in the observational follow-up cohort.
- Missing Data: Sensitivity analyses, including inverse probability weighting, were used to account for potential selection bias in the extension study (those who consented vs. those who did not), and results remained consistent.
- Event Adjudication: A significant strength is the use of an expert clinical panel for blinded adjudication of cancer types, stages, and causes of death, which reduces ascertainment bias.
Applicability
- Patient Population: The study is highly relevant to “healthy” older adults (mean age 75) who are considering primary prevention.
- Clinical Practice: These results suggest that for patients ≥70 years, the “aspirin-for-all” preventive strategy is likely harmful regarding cancer outcomes. Clinicians should prioritize other screening and prevention methods in this demographic.
Research Objective and Study Design
- Objective: To assess the 10-year association between low-dose aspirin and cancer incidence/mortality and identify potential “legacy effects” after stopping treatment.
- Design: A community-based binational (Australia/US) cohort study derived from the ASPREE randomized clinical trial (2010–2017) and its observational extension (ASPREE-XT, 2018–2024).
- Setting and Participants: 19,114 older adults (mostly ≥70 years) free of cardiovascular disease, dementia, or physical disability at baseline.
Bibliographic Data
- Title: Cancer Incidence and Mortality With Aspirin in Older Adults: Follow-Up of the ASPREE Trial
- Authors: Suzanne G. Orchard, PhD; Galina Polekhina, PhD; John Zalcberg, MBBS; et al.
- Journal: JAMA Oncology
- Year: 2026
- DOI: 10.1001/jamaoncol.2025.6196
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
