Comment:
The clinical management of advanced melanoma has been transformed by the arrival of anti-PD-1 immunotherapy, yet we frequently observe a frustrating divergence in response among patients receiving the same conventional standard-of-care. This observational cohort study of 200 patients identifies a critical metabolic requirement that likely dictates the success of these conventional treatments: serum vitamin D levels.
This study exposes the failure of treating advanced melanoma with anti-PD-1 agents in a state of vitamin D deficiency. Patients who entered treatment with low vitamin D levels saw their progression-free survival cut nearly in half compared to those with sufficient levels. This identifies a clear survival hazard that is entirely preventable. As naturopathic doctors, we understand that the structural integrity of CD8 lymphocytes—the very cells harnessed by immunotherapy—is influenced by the vitamin D prohormone.
Although the 4.5-month numerical advantage in overall survival did not reach statistical significance in this cohort, the doubling of progression-free survival from 5.75 to 11.25 months is a definitive clinical signal. Unfortunately there is no data on the levels of serum vitamin D achieved and whether that subgroup have even better outcomes, as we know from other studies that levels above 50 are necessary to see improvements in overall survival.
The definitive takeaway is that vitamin D testing and supplementation, something we almost certainly do, should be integrated as a standard procedure for all melanoma patients, and likely all patients, starting immunotherapy to ensure the system is capable of supporting a successful clinical outcome.
The Wonk Debate – Audio Critique & Clinical Commentary:
Summary:
Clinical Bottom Line
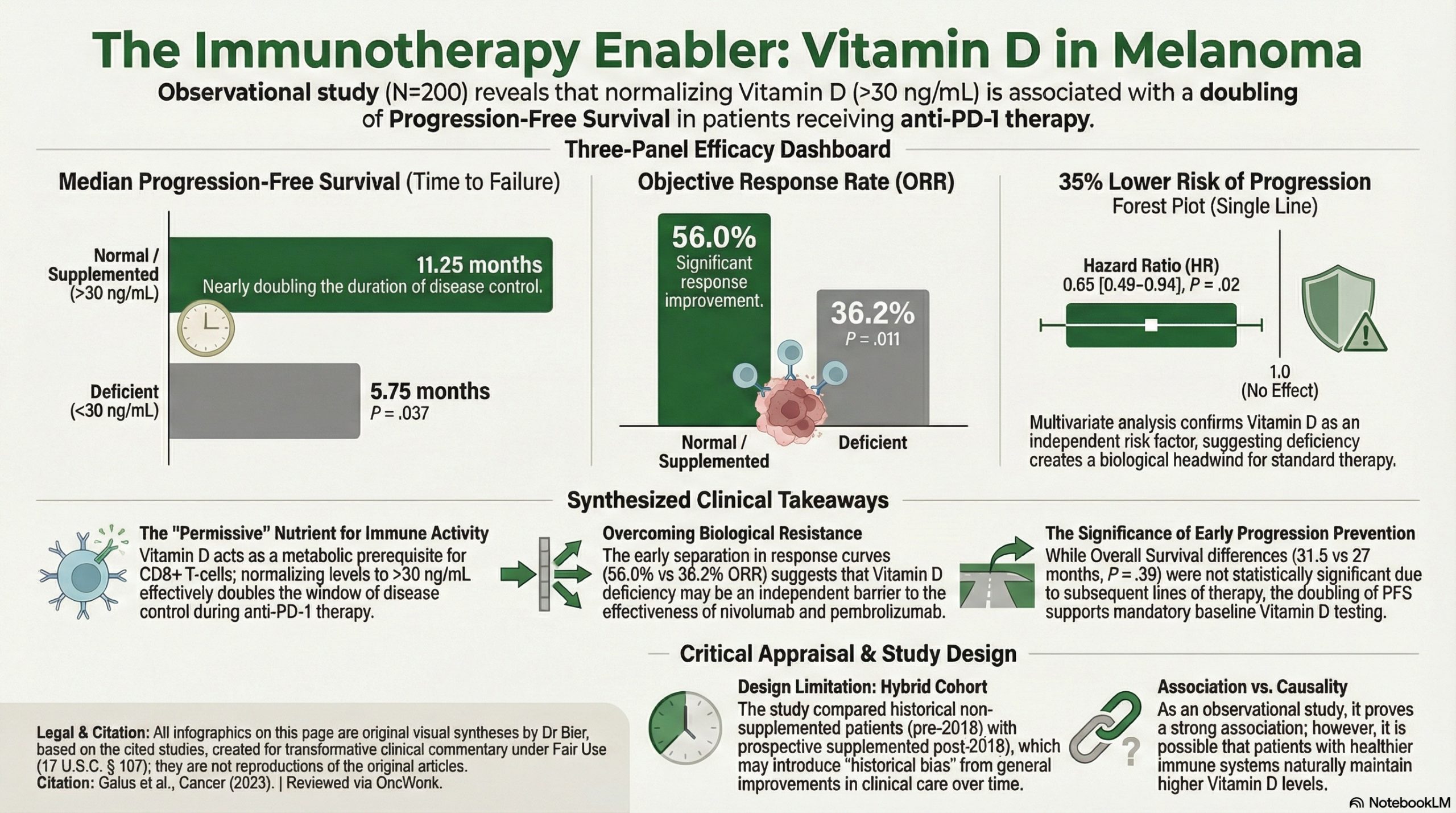
This observational study suggests a significant association between normal serum vitamin D levels and improved clinical outcomes in patients with advanced melanoma treated with anti-PD-1 immunotherapy. Patients with normal vitamin D levels (attained naturally or via supplementation) achieved a higher Objective Response Rate (56.0% vs. 36.2%) and nearly double the median Progression-Free Survival (11.25 vs. 5.75 months) compared to those with low levels. While these results are provocative and biologically plausible, the study’s non-randomized, partially retrospective design means it cannot prove that vitamin D supplementation causes better outcomes, as unmeasured confounding variables (e.g., overall health status) may be at play.
Results in Context
- Main Results:
- Objective Response Rate (ORR): 56.0% in the normal/supplemented group vs. 36.2% in the low vitamin D group (p = .011).
- Progression-Free Survival (PFS): Median PFS was 11.25 months in the normal/supplemented group vs. 5.75 months in the low group (p = .037).
- Overall Survival (OS): 31.5 months in the normal group vs. 27 months in the low group; however, this was not statistically significant (p = .39).
- Definitions: * Objective Response Rate (ORR): The proportion of patients with a reduction in tumor size of a predefined amount.
- Progression-Free Survival (PFS): The length of time during and after treatment that a patient lives with the disease, but it does not get worse.
- Hazard Ratio (HR): In the multivariate analysis, reduced vitamin D was an independent risk factor for progression (HR 0.65 for normal vs. reduced, p = .02), meaning patients with normal levels had a 35% lower risk of progression at any given time.
- Participants: A total of 200 patients were included: 58 in the reduced vitamin D subgroup and 141 in the normal/supplemented subgroup (one patient was excluded from analysis).
Assertive Critical Appraisal
- Limitations & Bias (STROBE Framework): * Confounding: As an observational study, the primary limitation is the potential for confounding. Patients with higher vitamin D levels might have higher performance status, better nutrition, or more outdoor activity, all of which could independently improve melanoma outcomes.
- Selection Bias: The study compared a retrospective cohort (pre-July 2018) who were not supplemented with a prospective cohort (post-July 2018) who were supplemented. This “historical control” approach introduces bias from changes in clinical practice over time.
- Reporting Quality Assessment (STROBE): The authors clearly describe their efforts to address confounding through multivariate Cox proportional hazards analysis, adjusting for factors like LDH levels, disease stage, and ECOG performance status. However, the lack of a fully prospective, randomized design is a major limitation that precludes a definitive causal statement.
- Applicability: Given the safety and low cost of vitamin D supplementation, these findings are highly relevant. In Poland, where the study was conducted, deficiency is estimated at 90% of the population, making this intervention broadly applicable to clinical practice in similar latitudes.
Research Objective
To compare the effectiveness of anti-PD-1 therapy (nivolumab or pembrolizumab) in patients with advanced melanoma in relation to their serum vitamin D levels.
Study Design
This was a hybrid retrospective-prospective observational cohort study. Patients treated before July 2018 were analyzed retrospectively using preserved serum; patients treated after July 2018 were managed prospectively with mandatory vitamin D testing and supplementation for those with levels ≥ 30 ng/mL.
Setting and Participants
- Setting: A single oncology center in Poznań, Poland.
- Eligibility: Patients with locally advanced, inoperable, or metastatic melanoma receiving first-line anti-PD-1 therapy.
- Exclusions: Patients with untreated CNS metastases.
Bibliographic Data
- Title: Vitamin D supplementation increases objective response rate and prolongs progression-free time in patients with advanced melanoma undergoing anti-PD-1 therapy
- Authors: Łukasz Galus, Michał Michalak, Mateusz Lorenz, et al.
- Journal: Cancer
- Year: 2023
- DOI: 10.1002/cncr.34718
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
