Comment:
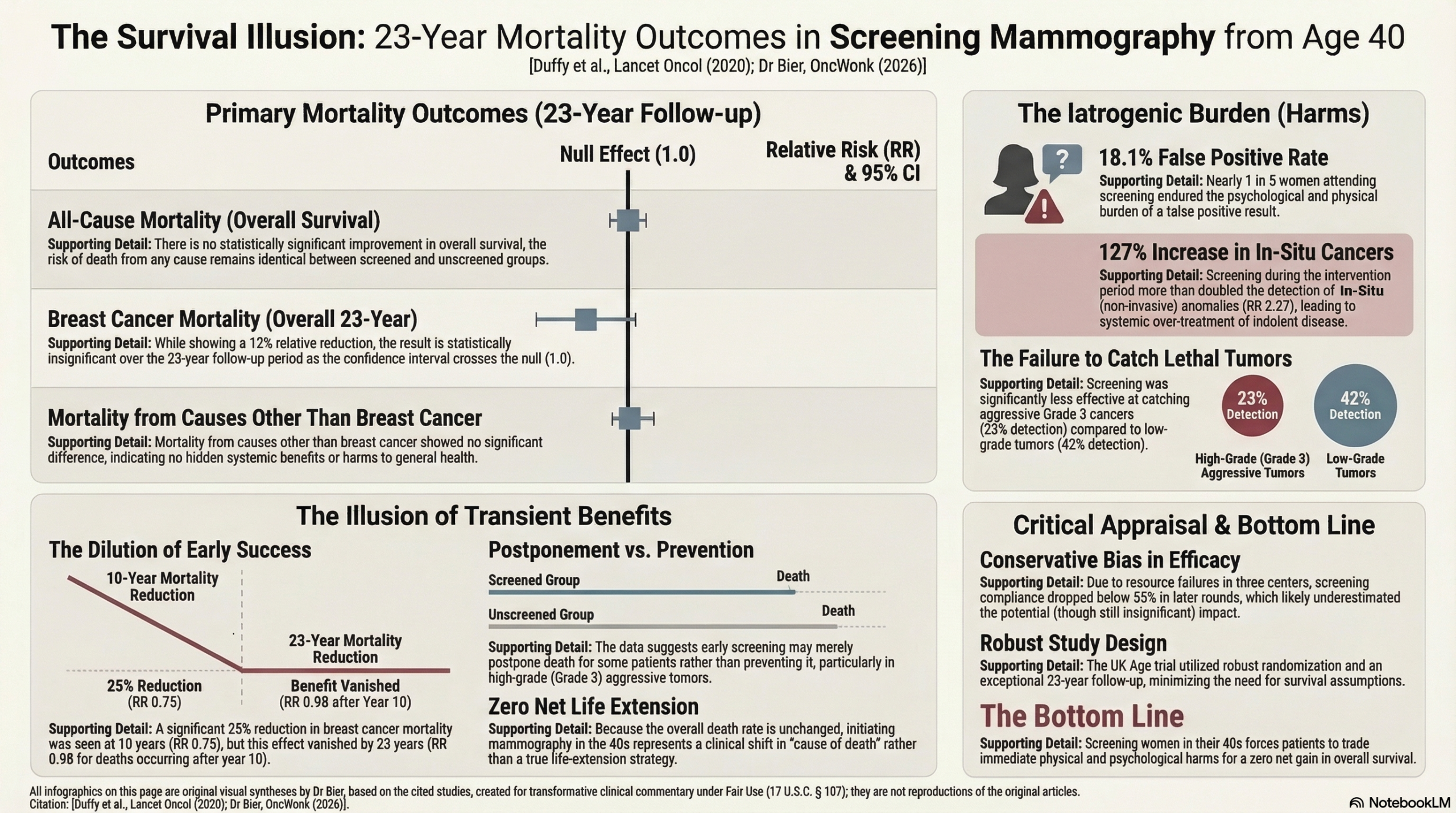
Finding more cancer is only a medical triumph if the patient actually survives longer as a result. This updated to the UK Age trial in women aged 40 to 49 shows that it fails to improve overall survival. While proponents focus on a temporary, early reduction in breast cancer mortality only (not overall survival), the 23-year follow-up reveals no significant long-term breast cancer benefit (RR 0.88) and zero improvement in all-cause mortality (RR 1.01).
This is always a difficult concept for people. If we lower deaths from one specific disease but the overall death rate remains identical, the intervention is causing iatrogenic harm that offsets the benefit. The trial exposes this harm plainly: 18.1% of screened women endured a false positive, and the detection of in-situ cancers more than doubled (RR 2.27) during the intervention period. We are systematically over-treating non-lethal anomalies. We do not currently have the technology that can successfully differentiate lethal from indolent disease, so screening women in their 40s currently serves as a Cause-of-Death shift on a spreadsheet rather than a genuine life-extending strategy.
The Wonk Debate – Audio Critique & Clinical Commentary:
Summary:
Clinical Bottom Line
This 23-year follow-up of the UK Age trial indicates that while annual mammographic screening for women in their 40s leads to a significant reduction in breast cancer-specific mortality during the first 10 years of follow-up (RR 0.75), this benefit is transient and does not translate into a statistically significant reduction in breast cancer mortality over the full 23-year period (RR 0.88). Most critically, there was no improvement in overall survival (all-cause mortality). For the general clinician, this suggests that while early screening may catch some aggressive cancers sooner, it does not change the ultimate survival trajectory for the population of women starting at age 40, likely because breast cancer deaths are a relatively small component of total mortality in this age group.
Results in Context
Primary Outcome: Breast Cancer Mortality
- The overall rate ratio (RR) for breast cancer mortality was 0.88 (95% CI 0.74–1.04) for tumors diagnosed during the intervention phase.
- Statistical Definition: An RR of 0.88 suggests a 12% relative reduction in the risk of breast cancer death in the intervention group compared to the control group. However, because the 95% confidence interval crosses 1.0 (0.74 to 1.04), this result is not statistically significant over the long term.
Oncology Endpoints & Response Criteria
- Overall Survival (OS): In the context of a screening trial, all-cause mortality serves as the most reliable measure of overall clinical benefit (Overall Survival). This study found an RR of 1.01 (95% CI 0.96–1.05) for all-cause mortality.
- Clinical Significance of OS: Although the trial was specifically powered to detect differences in breast cancer-specific mortality, the lack of an OS benefit is a critical finding. OS is the gold standard for clinical benefit because it accounts for both the potential life-saving effects of the intervention and any potential harms (e.g., treatment-related deaths).
- Disease-Specific Mortality (Surrogate context): The significant reduction in breast cancer mortality seen in the first 10 years (RR 0.75, 95% CI 0.58–0.97) suggests an early benefit that “dilutes” as the control group begins routine screening at age 50.
Harms and Adverse Events
- Overdiagnosis: The study found no evidence of substantial long-term overdiagnosis of invasive cancers.
- In-situ Disease: There was a significant increase in the incidence of in-situ (non-invasive) cancers in the intervention group (RR 1.36, 95% CI 1.11–1.67) when including cancers detected at the first routine screen at age 50.
- False Negatives: 23% of grade 3 (highly aggressive) cancers were detected via screening, whereas 42% of grade 1 and 2 cancers were screen-detected, suggesting that screening is less effective at catching the most lethal, fast-growing tumors early enough to prevent death.
Assertive Critical Appraisal
Risk of Bias (RoB 2 Framework)
- Judgment: Some concerns.
- Critique: While the individual randomization was robust and stratified by GP practice, three of the 23 centers had to cease screening early due to insufficient resources. By the seventh screening round, less than 55% of women in the intervention group were actually being screened, which likely led to an underestimation of the intervention’s potential efficacy (a “dilution” of the effect).
Subgroup Analyses (Time Periods)
- The authors highlight a significant mortality reduction in the first 10 years.
- Appraisal: While this period-specific effect is statistically significant (RR 0.75), the clinician should view it with caution. This “early win” did not persist. The lack of a long-term significant effect suggests that early screening might be postponing death rather than preventing it, particularly for high-grade (Grade 3) tumors.
Reporting Quality Assessment (CONSORT)
- Quality: High.
- Assessment: The paper includes a clear participant flow diagram. It adequately describes the randomization methods (computer-generated) and allocation concealment . The primary analysis followed the intent-to-treat (ITT) principle, which is essential for preserving the benefits of randomization.
Applicability
- The trial reflects the UK NHS environment of the 1990s. Modern digital mammography and two-view screening (this trial used single-view for subsequent screens) have higher sensitivity and would likely yield better detection rates today. However, the fundamental finding that screening in the 40s does not improve overall survival remains a highly generalizable and sobering caution for clinical practice.
Research Objective and Study Design
- Objective: To compare the effect of annual mammographic screening starting at age 40 versus starting at age 50 on breast cancer mortality.
- Design: Individual randomized controlled trial (1:2 allocation).
- Participants: 160,921 women aged 39–41 years recruited from 23 UK Breast Screening units.
- Intervention Group (n = 53,883): Offered annual mammography until age 48.
- Control Group (n = 106,953): Received usual care (first invitation at age 50).
Bibliographic Data
- Title: Effect of mammographic screening from age 40 years on breast cancer mortality (UK Age trial): final results of a randomised, controlled trial
-
Authors: Stephen W Duffy, Daniel Vulkan, Howard Cuckle, Dharmishta Parmar, Shama Sheikh, Robert A Smith, et al.
-
Journal: The Lancet Oncology
-
Year: 2020
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
