Comment:
We should assess risk and benefits of any intervention not only by the physical impact, but by the measurable psychological damage it inflicts. The claims data analysis highlights the conflict between the medical establishment’s routine dismissal of a false-positive mammogram as a benign inconvenience and the structural reality that it actively drives patients toward initiating psychiatric medications.
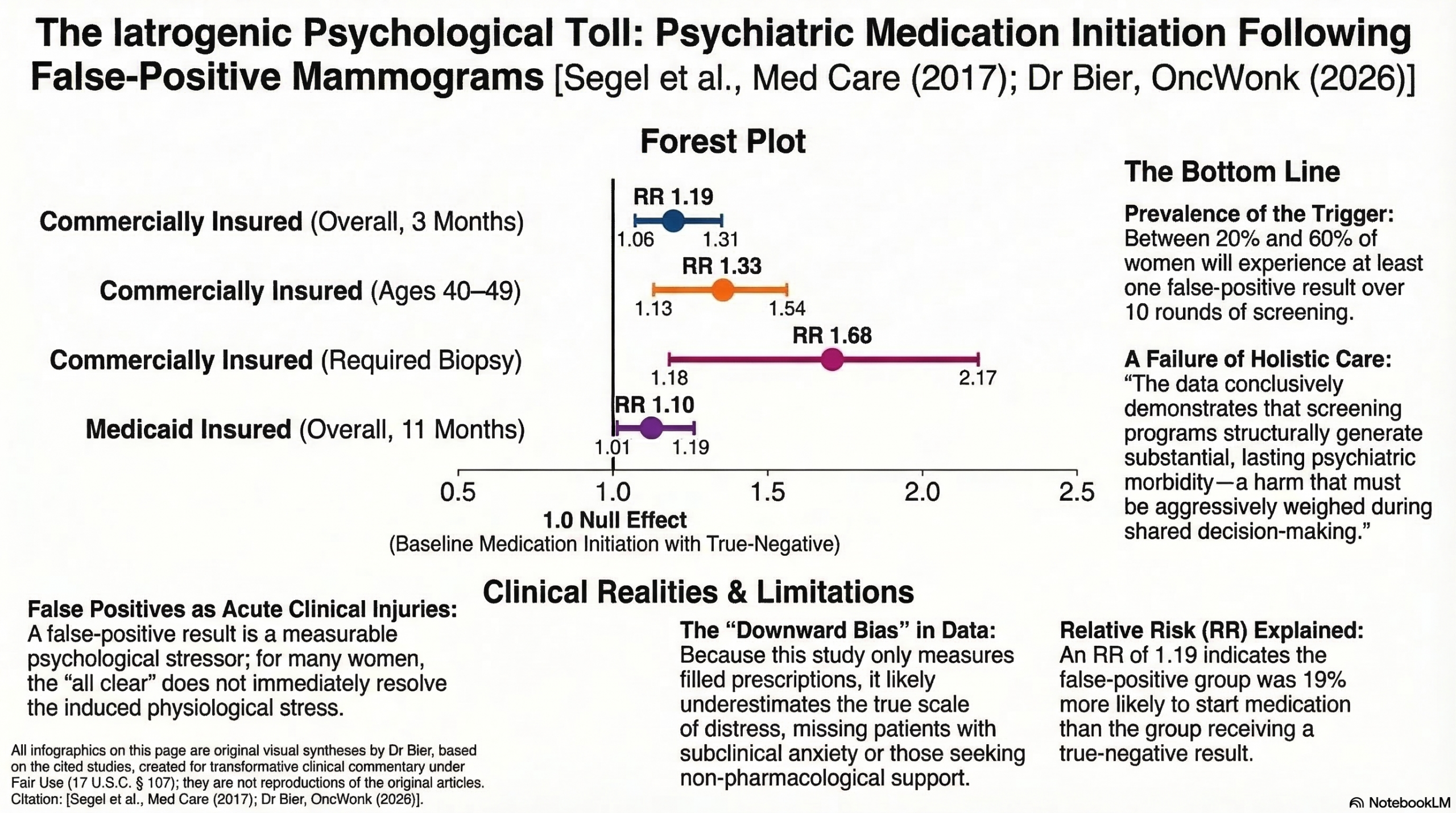
We must acknowledge how incredibly common this trigger is: data demonstrates that after 10 rounds of screening, between 20% and 60% of women will experience at least one false alarm. When this happens to younger, commercially insured women aged 40 to 49, they face a 33% increased risk (RR 1.33) of initiating antidepressants or anxiolytics. Even across the broader commercial cohort, there is a 19% increased risk (RR 1.19) within three months. Crucially, the systemic harm scales with the duration and invasiveness of the scare: when diagnostic resolution takes longer than a week, the risk climbs (RR 1.21), and if a biopsy is required, even when it comes back benign the risk of psychiatric medication initiation skyrockets by 68% (RR 1.68). The severe physiological stress provoked by an existential cancer threat does not simply evaporate when the “all clear” is given; it creates a lasting psychological wound. We must recognize false positives as acute, measurable clinical injuries, and take that into account in our analysis of the risks of screening mammography.
The Wonk Debate – Audio Critique & Clinical Commentary:
Summary:
Clinical Bottom Line
This retrospective cohort study provides evidence that false-positive mammograms are associated with a modest but statistically significant increase in the initiation of antidepressant or anxiolytic medications within three months of the screening event. The risk is most pronounced in commercially insured women aged 40–49 and those whose follow-up required more invasive or prolonged procedures, such as a biopsy or multiple tests. While observational data cannot definitively prove causation, these findings suggest that the psychological distress following a false-positive result reaches a clinically actionable level for a subset of patients, highlighting the need for rapid resolution of suspicious findings and targeted mental health monitoring.
Results in Context
Main Results
The study identified a significant increase in medication initiation following a false-positive result compared to a negative result, though the magnitude varied by insurance type and clinical characteristics:
- Commercially Insured Population: At 3 months, the relative risk (RR) for initiating an antidepressant or anxiolytic was 1.19 (95% CI, 1.06–1.31). By 11 months, the risk remained elevated but slightly lower at 1.09 (95% CI, 1.02–1.16).
- Medicaid Population: The 3-month risk was 1.13 (95% CI, 0.96–1.29), which was not statistically significant. However, by 11 months, the RR was 1.10 (95% CI, 1.01–1.19), reaching statistical significance.
- High-Risk Subgroups (Commercial):
- Age 40–49: RR 1.33 (95% CI, 1.13–1.54).
- Involved Biopsy: RR 1.68 (95% CI, 1.18–2.17).
- Multiple Follow-up Tests: RR 1.37 (95% CI, 1.17–1.57).
- Resolution >1 Week: RR 1.21 (95% CI, 1.07–1.34).
Definitions
- Relative Risk (RR): A measure of the probability of an event (medication initiation) occurring in the exposed group (false-positive) compared to the unexposed group (negative). An RR of 1.19 means the false-positive group was 19% more likely to start medication than the negative group.
Participants
The study analyzed a substantial cohort of women aged 40–64 with no prior history of antidepressant or anxiolytic use:
- Commercially Insured: 85,522 total women, including 8,445 with false-positives.
- Medicaid Insured: 32,268 total women, including 3,665 with false-positives.
Assertive Critical Appraisal
Limitations & Bias (STROBE Framework)
- Misclassification Risk: Because the study relied on claims data rather than medical records, mammogram results were inferred using a validated algorithm. While the algorithm has high specificity (99.4%), its sensitivity is 74.9%, which likely biased the results toward the null, suggesting the true effect may be larger than reported.
- Selection Bias: The requirement for continuous enrollment (12 months before and 9 months after the mammogram) may limit generalizability, particularly for the Medicaid population where “churning” in and out of eligibility is common. This likely resulted in a “healthier” or more stable sample than the general Medicaid population.
- Unmeasured Confounding: While the authors adjusted for age, insurance type, and comorbidities, they lacked data on race/ethnicity for the commercial sample and other clinical factors like hormone therapy use, which could influence both mammogram accuracy and baseline anxiety.
Reporting Quality Assessment (STROBE)
The authors clearly described their efforts to address potential confounding by using multivariate logistic models and controlling for several individual-level variables. They also conducted sensitivity analyses—such as looking only at the first observed mammogram and varying continuous enrollment requirements—which reinforced the robustness of their findings.
Reporting Quality Assessment (RECORD) for RWE Studies
The study adequately describes the data sources (MarketScan Commercial and Medicaid databases). The definitions for exposures (false-positive mammograms) and outcomes (medication initiation via therapeutic class codes) were explicitly stated and based on previously validated approaches.
Applicability
The findings are highly relevant to primary care and radiology settings in the U.S.. The results underscore that the “harm” of a false-positive is not merely financial or a temporary inconvenience but can lead to long-term clinical psychiatric intervention.
Research Objective
To analyze the effect of a false-positive mammogram on the initiation of antidepressant or anxiolytic medications and to identify specific subpopulations at elevated risk for these adverse psychological outcomes.
Study Design
This was a retrospective cohort study using large-scale commercial and Medicaid insurance claims databases.
Setting and Participants
- Setting: U.S.-wide claims data from medium/large self-insured employers (Commercial) and eight geographically diverse states (Medicaid).
- Dates: 2003–2004 for commercial data; 2003–2007 for Medicaid data.
- Eligibility: Women ages 40–64 undergoing screening mammography with no prior history of antidepressant or anxiolytic claims in the 12 months preceding the screen.
Bibliographic Data
- Title: The Effect of False-positive Mammograms on Antidepressant and Anxiolytic Initiation
- Authors: Joel E. Segel, PhD; Rajesh Balkrishnan, PhD; Richard A. Hirth, PhD
- Journal: Medical Care
- Year: 2017
- DOI: 10.1097/MLR.0000000000000755
Fair Use & Copyright: This post provides a transformative, thesis‑driven critical appraisal intended for educational and scholarly purposes. It is not a reproduction of, nor a market substitute for, the original research article.
Support the Version of Record: To support the copyright holders and verify the underlying data—including primary survival curves, risk estimates, and other core outcomes—readers are strongly encouraged to access the original Version of Record via the link or DOI provided above.
Medical Disclaimer: This content is for informational and educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment.
